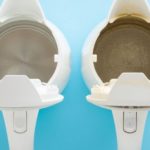
On average, each British tea lover consumes 2.5 kg of tea per year and the UK population drinks 70 million cups of coffee per day! That’s a lot of boiling kettles creating a lot of limescale over time.
It’s pretty easy to combat limescale, with the option of descaling tablets which you can purchase from most supermarkets, but if you prefer to try natural methods, then a vinegar and water solution could be your new go-to!
While most people recommend using white vinegar as it has less flavour and smell, you can also use malt vinegar to descale your kettle. You will just want to rinse the kettle out well afterwards (or you could enjoy a new flavour of tea – chamomile and malt vinegar anyone!?).
You’ll need:
- Malt vinegar
- Water
It’s that simple!
With only 2 necessary components; one available from your local shop and the other sourced directly from your tap at home, there could not be an easier way to keep your kettle sparkling and clean.
How to Descale a Kettle with Malt Vinegar:
- Pour vinegar into the kettle to cover the elements plus a little bit
- Leave it for an hour or so
- Add just over an equal amount of water to the vinegar (or more if you like)
- Boil the kettle
- Pour away the vinegar solution and admire your newly descaled kettle!
Optional tip:
Rinse out your kettle a couple of times, or boil plain water to avoid any vinegar smells or tastes being left behind. Use the boiled water to wash some dishes to avoid wasting it! The hint of vinegar could even help to cut through grease on baking trays or frying pans.

Natural AND Cheap
Using vinegar to descale your kettle is so much better for the environment than chemical tablets and it’s also a heck of a lot more friendly on your purse or wallet.
Clean your kettle regularly to keep on top of limescale by filling it with water to just below the max line then adding 3 tbsp of vinegar before boiling. This can be done every couple of months, or whenever you think it needs it.
Adding water is sometimes not even necessary; you could try popping the vinegar in to cover the elements and just leave it for a few hours or overnight. The vinegar will work it’s magic and your kettle will be clean as a whistle – no boiling necessary.
Just make sure to warn any early-risers who come to make a sleepy cuppa first thing in the morning, or they might get a bit of a malty shock…
Important tip:
Make sure you use more water than vinegar or you’ll find that the vinegar causes the solution to splash out of the spout.


In The Wash is your guide to the best laundry and cleaning products, tips and tricks. Our mission is to solve the UK’s cleaning and laundry dilemmas!